 
Supercritical fluid impregnation involves delivering the active substance onto an inert substrate, In its gaseous state, the CO2 leaves the target substance impregnated with the active substance. Supercritical fluids can be used to deactivate or destroy some bacteria, fungi, and yeasts through the high pressure disrupting or destroying the cell walls They are therefore used in sterilising food and medical instruments. Once dissolved in supercritical CO 2 depressurisation releases the extracted components and the CO2 is returned to a gas This can then either be evaporated harmlessly into the air, or recycled by condensation. The main advantage of supercritical carbon dioxide (CO 2) has over solvents used in extraction processes its high diffusion and the ability to selectively adjust the properties to selectively extract different components. Here are more details about some of the major processes involving supercritical fluids. How do supercritical fluid processes work? This can be useful for synthetic alternations such as hydrogenations. This leads to much higher concentrations of dissolved gases, compared to the effects achieved using solvents. Supercritical fluids also mix readily with gases, such as nitrogen or hydrogen. creating finely divided solids in the nano range. This rapid precipitation prevents material agglomeration or crystal growth. By dissolving a substrate in the supercritical phase then rapidly expanding the contents through depressurisation the dissolved materials rapidly precipitate. In addition to extraction, SCF can be used to micronise materials. 
This solubility is an essential part of the polymerisation process. Supercritical fluids can perform various processes, including:įor most solutes, supercritical Carbon dioxide solvent power is similar to that of light hydrocarbons, such as hexanes and pentanes. In fact, fluorinated compounds are more soluble in supercritical CO 2 than in hydrocarbons. What are the uses of supercritical fluids? By varying the temperature and pressure between 150☌ and 350☌, and 15 to 200 bar the properties of the fluid can be alternated to selectively extract natural materials. The subcritical region of water is highly compressed water that is still in its liquid state. However subcritical water is commonly used as an equivalent or alternative to supercritical CO2. Water has a higher supercritical point than CO 2, at 374☌ and 220 bar but it is well-suited to various hydrothermal processes, for instance oxidation in waste processing (wet air oxidation, or WAO) and processes to treat hazardous waste and synthesise nanoparticles.Īt supercritical levels, oxidation reactions occur, and organic compounds and gases will mix together homogenously. Subcritical and Supercritical water (H 2O)? This means you can use supercritical CO 2 fluid at mild temperature conditions, between 40° and 60☌, and it will not leave behind harmful organic residues. Low-cost while being available at high levels of purity.This coupled with the following properties makes it one of the most versatile green solvent available: Supercritical CO 2 has the most accessible critical temperature and pressure at 31☌, 74 bar. Other supercritical fluids can be created from the following substances: With Carbon dioxide and water being the most commonly used supercritical fluid used within industrial applications. diffusing through solids like a gas, while.These properties create supercritical fluids that can be manipulated so be used in industrial processes by diffusion and viscosities exhibited by gases.densities equivalent to that of liquids.Supercritical fluids, or SCFs, create fluids with: What are the properties of supercritical fluids? 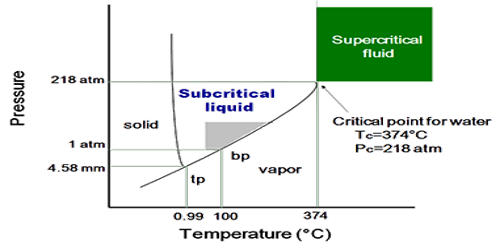
Further manipulation of these conditions can be used to create a variety of unique properties used to extract a number of natural products. High diffusion like a gas but with the solvation (ability to dissolve substances) of a liquid. This is achieved by increasing the temperature or pressure.įor example, increasing the temperature above 31☌ and pressure above 73 bar for Carbon dioxide creates a supercritical phase, neither liquid nor gas but a combination of both properties. The properties of this fluid can be adjusted by going further into the supercritical region. How do you create a Supercritical Fluid?Īpplying temperature and pressure above the critical point of a substance, pushes that substances into the supercritical phase. They form the basis of clean technology as an alternative solvents for extracting natural products, chemicals and other substances. Supercritical fluids have a range of supporting technologies used in several industrial applications. A supercritical fluid is a highly-compressed fluid that combines the properties of gases and liquids. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
